Vaccines are great, we all know this! But how exactly are they developed? Surely it’s not as simple as it sometimes seems. Well it isn’t. There are many steps and protocols to follow before vaccines are just injected into the public. These steps include the exploratory stage, pre-clinical stage, and clinical development, which includes three stages in itself. In order to get more vaccines in the future, such as one for HIV, these protocols must be followed, which will be discussed in today’s post. Let’s remember, since vaccines do take serious work to develop, and since they protect against contractible diseases, GET VACCINATED when you can!
According to the CDC, this list of protocols to get a vaccine prepared can take 10-15 years in some cases. In an article by the CDC they break up the stages and explain them, so these 10-15 years are more comprehensible. The first two steps of vaccine development involve laboratory and animal testing (poor mice). In the first exploratory stage, scientists work to find an antigen that has the potential to prevent against a specific disease, whether these are natural or synthetic antigens. This stage can last anywhere for 2-4 years, which makes sense considering they have to find ONE antigen to prevent an entire, and sometimes evolving DISEASE. The pre-clinical stage uses culture and animals to see if there is an immune response, and if this reaction is safe. This stage generally lasts for 1-2 years. After this, an application must be submitted to the FDA, and they have 30 days to review and either approve or deny the application. That sounds slightly intense! Lastly in the creation process, there are three stages of clinical development, which can only happen once the FDA has approved the proposed new vaccine. After animal studies have been done, it is time for human trials. The first stage of clinical development involves human studies, and the main goal is to understand the immune response in humans and safety of this vaccine. The trial is a small sample size, generally 20-30 people. I hope they are getting paid good money to participate in this! The second stage has similar goals, but includes are larger sample population. This stage also looks at mode of delivery of the vaccine, as well as the type of schedule it should have. The last stage before approval looks to see if there are any rare side effects, doing so by looking at a much larger group, generally thousands. A larger sample size helps scientists understand if the vaccine actually works and if there is anything they need to alter that could cause side effects. After successful clinical studies, this vaccine can now move on to getting officially approved. Now enough of the logistics, let’s talk about a vaccine that is attempting to be developed in real life!
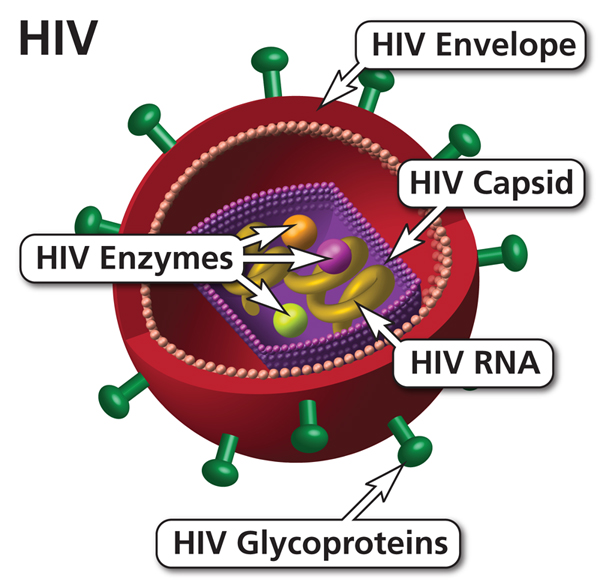
Let’s talk about an article that was released just FOUR DAYS AGO! Isn’t it crazy how constantly science changes?! It’s insane! This article discusses what is going on with the development of an HIV vaccine. Due to HIVs changing and mutating nature, scientists are having to take a different approach with this vaccine than they have with others. They use a piece of protein to attract rare B cells, which multiply once binded to the protein. By attracting these rare B cells, scientists believe they will be able to more broadly neutralize antibodies, which are necessary with a changing disease. So why has this wonderful vaccine not been released yet? Well as shown before, it is a long and grueling process. This past year scientists have been working to find the necessary rare B cells. They tested in mice and found a B cell that has an antibody that helps prevent HIV in mice. That’s amazing! There is still a long way to go with this vaccine, but I cannot wait to see how it develops over the coming years.